Can gold "generate" itself? Chinese scientists have for the first time captured the formation mechanism of nano gold, potentially rewriting mining rules
A Chinese Academy of Sciences team has captured, for the first time using in-situ electron microscopy, the entire process of gold “growing” on the surface of pyrite, bringing new possibilities for low-temperature, low-pollution gold extraction techniques.
(Background: Will precious metals still be purchasable in 2026? Analyzing why gold has become the best target for the US midterm election “Trump trade”)
(Additional context: Is gold price about to explode? HSBC predicts: Gold will surge to $5,000 in the first half of 2026)
Table of Contents
- Real-time recording of gold “growth”
- Discrepancy between chemical mechanisms and traditional beliefs
- Potential implications for mining and ESG
- Next steps await industrial validation
Gold has recently hit record highs repeatedly, with investor sentiment remaining hot. An interesting piece of news is that a team from the Chinese Academy of Sciences and Guangzhou Institute of Geochemistry published research capturing the full process of gold formation on pyrite surfaces in aqueous solutions, sparking discussions in the mining and commodities markets.
Real-time recording of gold “growth”
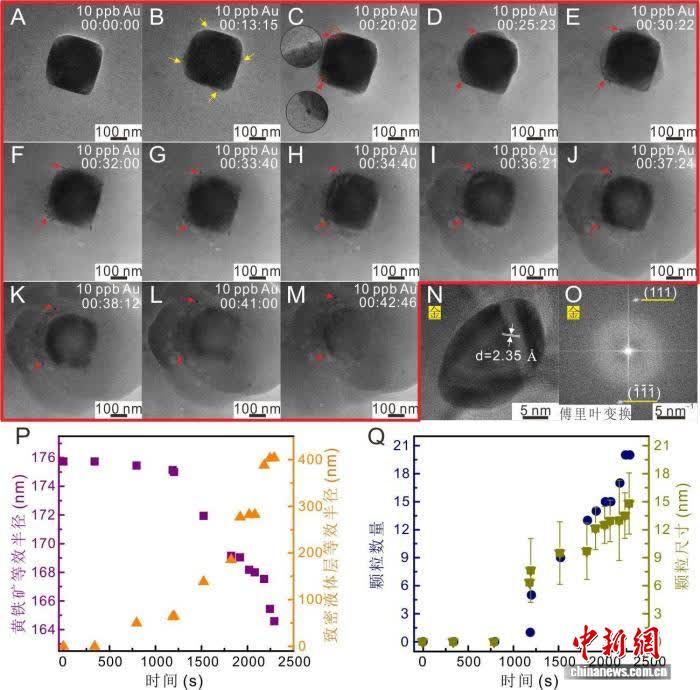
Researchers used in-situ liquid-phase transmission electron microscopy, placing a gold-containing solution with a concentration of only 10 ppb together with pyrite crystals in a sealed observation chamber.
At the 13-minute mark, the camera captured a dense liquid layer forming on the crystal surface; by 20 minutes, gold nanoparticles began nucleating and aggregating within that layer. The experiment first demonstrated that, without high temperature or pressure, under near-surface mild conditions, pyrite can promote the reduction of gold ions, resulting in metallic gold.
Image source: Guangzhou Institute of Geochemistry, Chinese Academy of Sciences
Discrepancy between chemical mechanisms and traditional beliefs
According to Wen Wei Po, the key lies in the fact that when pyrite dissolves, it consumes a large amount of oxygen molecules in the solution, causing the local oxygen fugacity to drop rapidly. The oxygen-deficient environment forces gold ions to gain electrons, precipitating as metallic gold.
This result challenges the traditional view that “gold deposits must originate from deep-seated hydrothermal systems,” explaining why fine-grained gold is often found coexisting with pyrite in riverbeds or surface weathered zones.
Potential implications for mining and ESG
Historically, gold extraction relied heavily on cyanide leaching, which is energy-intensive and toxic, raising concerns among ESG investors. The interface chemical reactions revealed by this study provide a theoretical basis for developing low-temperature, low-toxicity gold leaching processes.
If the microenvironment of the dense liquid layer can be artificially simulated, low-grade ores and tailings piles might be able to recover precious metals without using highly toxic reagents, reducing costs and improving environmental friendliness.
Next steps still require industrial validation
Although the experiments used extremely low concentrations close to natural water bodies, scaling up to industrial levels still faces challenges such as reaction rates, liquid control, and costs. The research team has not yet announced plans for mass production. However, this achievement at least proves the concept that “shallow, low-temperature conditions can also produce gold,” opening new research directions for global gold mining and recycling.